Most people don't need another blood test. They need someone to explain the one they already paid for.
That's the opportunity, and it's quieter and more defensible than the noise around longevity clinics, biohacking memberships, and premium preventive testing. Quest and Labcorp already run the labs. Annual physicals already produce the results. Employer screenings already push PDFs into inboxes. What's missing is the layer between the lab report and the next doctor appointment: a polished, plain-English interpretation that costs less than dinner and reads like a professional document instead of a chatbot answer.

The wedge is deliberately narrow. A $29 educational bloodwork report that turns a standard Quest or Labcorp PDF into a structured lifestyle roadmap, anchored in mainstream citations and framed around the functional-medicine tradition of "optimal" ranges. No lab draw. No membership. No promise to replace a clinician. A one-shot trust product built for the 35- to 60-year-old who got results back, googled three markers at midnight, and wants a calmer second opinion before their next visit.
Here's the opportunity:
The money: 500 reports a month is roughly $14,500 MRR. 1,000 is ~$29,000 MRR, or $348K annualized, run by two people.
Inside:
• MVP scope: the 30-50 biomarkers that matter
• Five-layer trust moat incumbents keep underbuilding
• Pricing ladder from $29 one-shot to B2B white-label
• SEO + Reddit playbook for lab-anxious searchers
The demand is already priced in
Grand View Research sizes the U.S. blood testing market at $35.9 billion in 2024, projected to reach $71.4 billion by 2033 at an 8.1% compound annual growth rate. This isn't a speculative category. It's an industrial diagnostic testing engine producing millions of confusing lab PDFs every week. McKinsey's 2024 research puts U.S. consumer wellness spend at roughly $500 billion, with 84% of Americans calling wellness a top or important priority and at-home diagnostics flagged as a durable trend line underneath the growth.
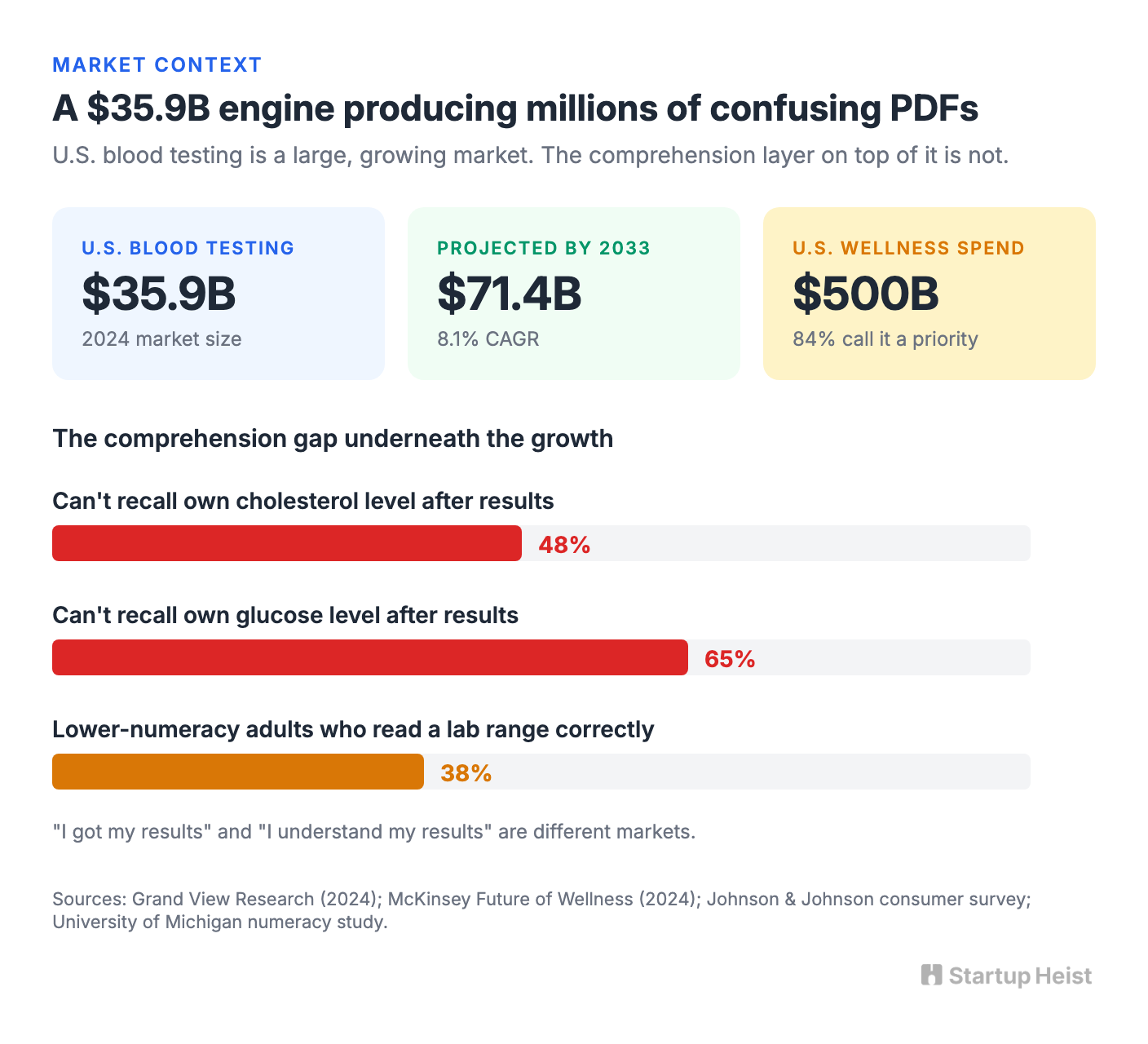
The piece that gets glossed over is the comprehension gap. A Johnson & Johnson survey captured it bluntly: 48% of Americans couldn't recall their own cholesterol level and 65% couldn't recall their glucose level after getting results back. A University of Michigan study found that only 38% of adults with lower numeracy skills could correctly tell whether a result fell outside the reference range. "I got my results" and "I understand my results" are different markets, and the second one is where the money is.
Why "AI reads labs" is the wrong framing
The obvious reaction is to build ChatGPT for lab reports. That market is already crowded, and the crowd reveals the opening.
Function Health sells a $365 annual membership for 160+ biomarkers with clinician review, dropped from $499 to widen the funnel. InsideTracker offers a $149 membership with a $340 Ultimate panel for members. Superpower charges $199 for 100+ biomarkers and an AI concierge layer, though user reviews note its recommended supplement stacks can run $400+ per month on top of the membership. SiPhox Health runs the aggressive low end: a free AI analyzer called Sai that accepts Quest and Labcorp PDFs in under a minute, bolted onto a $125 testing membership. Practitioner tools like LabSmarts, OptimalDX, and Functional DX supply functional-range lab interpretation to clinicians.
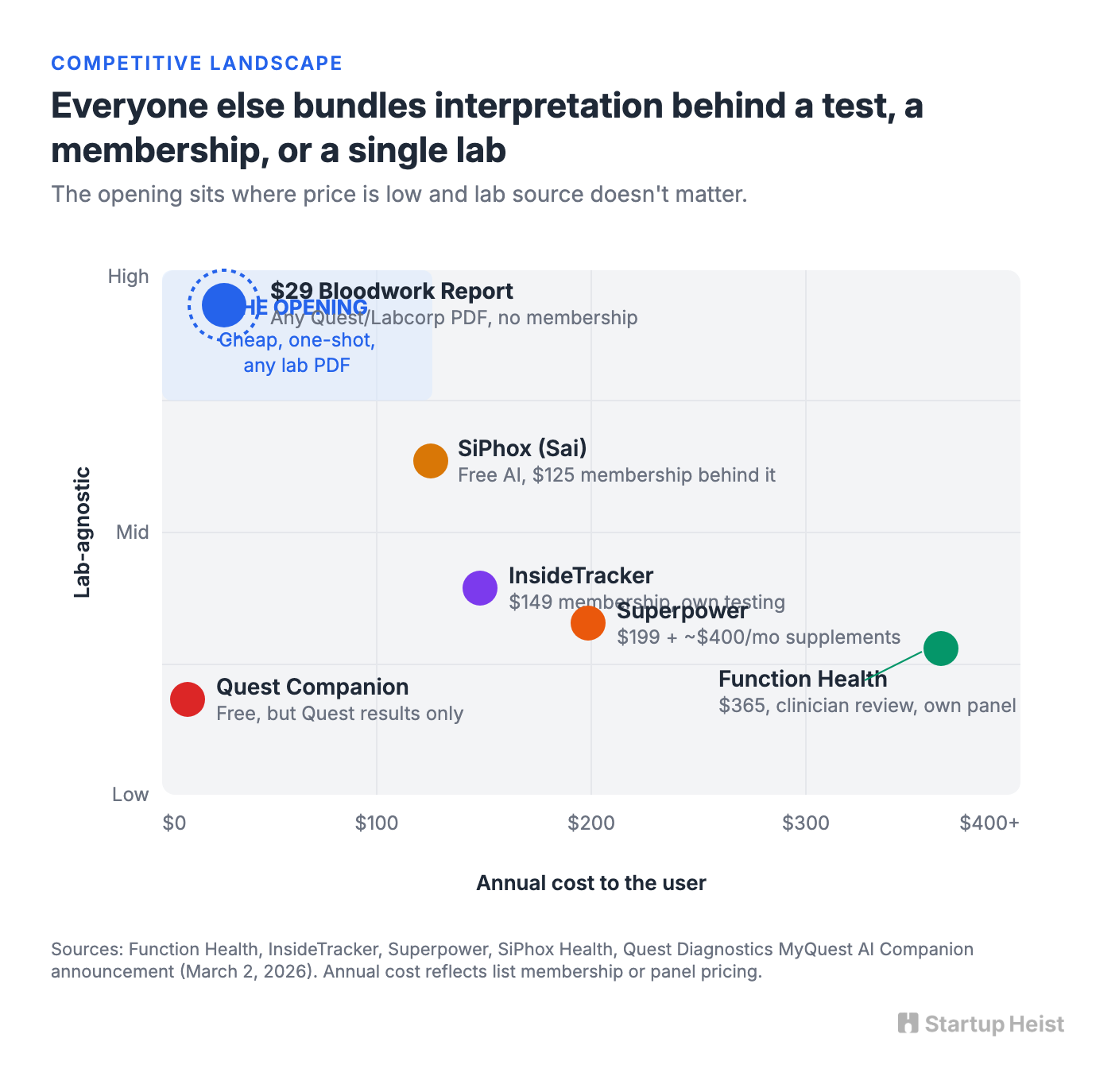
On March 2, 2026, Quest quietly raised the ceiling. MyQuest now ships an AI Companion powered by Google Gemini, free to every adult user, with access to up to five years of Quest lab history. That's a well-resourced, directly-integrated player in the space, and it's the one most people would point to as the "why bother" objection.

It actually sharpens the opening instead of closing it. Quest's Companion lives inside Quest and only reads Quest results. Function, InsideTracker, Superpower, and SiPhox all bundle interpretation into their own testing product, membership, or coaching layer. Across every one of these players, interpretation arrives late, costs more, drags a subscription behind it, or only works if you already used that specific lab. The opening is downstream and lab-agnostic: the user who will never pay $365 for Function, whose annual panel was run through Labcorp at their PCP's office, and who opens a Quest Companion account only to realize their own report isn't in it. That user doesn't need another test or another portal. They need a serious-feeling document that walks through the PDF they already have, regardless of who drew the blood.
Where the defensibility actually lives
At the low end, the winner is whoever lets you upload a PDF fastest. At the top end, the winner is whoever a nervous consumer is willing to pay for, immediately, after opening a confusing report at 11 p.m. A chatbot answer looks disposable because it is. A 20-page PDF with an executive summary, out-of-range markers, plain-English explanations, cited sources, a restrained supplement section, a "questions for your doctor" appendix, and a bright red escalation page for critical findings feels like a product. That perception gap is worth $29 in a category where the alternative is guessing.

Trust is also where the regulatory line gets drawn, and the 2026 guidance made it sharper. On January 6, 2026, the FDA issued updated final Clinical Decision Support Software guidance, replacing the September 2022 version. The update clarifies which software functions are excluded from the "device" definition under section 520 of the Food, Drug and Cosmetics Act, and places new emphasis on transparency around data inputs, underlying logic, and how AI-driven recommendations are generated. It also flags automation bias, meaning human over-reliance on machine suggestions, as a design concern the FDA now cares about. HIPAA exposure runs parallel. Route PHI through HIPAA-eligible endpoints under a signed Business Associate Agreement (AWS, Anthropic via AWS Bedrock or Google Cloud or Azure, OpenAI via qualifying API plans with zero data retention) or don't handle it at all.
The design rule is simple. The product produces educational and nutritional guidance generated from user-supplied lab data, with transparent citations and repeated instructions to consult a licensed clinician for diagnosis, treatment, medication changes, or urgent abnormalities. Hold that line and this is a legitimate micro-SaaS. Drift into quasi-diagnostic claims and it turns into a regulatory headache.
Why this can still win in a crowded field
SiPhox wins on free speed. Function wins on biomarker coverage and clinician review. Superpower wins on AI concierge framing, then gives that trust back every time the recommended supplement list crosses $400 a month. Quest's Companion wins on integration, but only for Quest customers.
None of them win on the one thing the confused 42-year-old wants at the moment of anxiety: a lab-agnostic document that feels authored, cited, and finished. That's the trust layer, and it rests on five pieces of infrastructure that are easy to underbuild:

Unlock the Vault.
Join founders who spot opportunities ahead of the crowd. Actionable insights. Zero fluff.
“Intelligent, bold, minus the pretense.”
“Like discovering the cheat codes of the startup world.”
“SH is off-Broadway for founders — weird, sharp, and ahead of the curve.”























