If you're hunting for healthcare startup ideas or B2B SaaS opportunities in surgical workflows, this one has an unusual structure: the hardest part — getting a new drug class approved, funded, and covered — is already done. Your job is to build the operating system around it.

A new category is opening up in surgical recovery, and the company that will own it won't be a pharma company. It will be the team that builds the workflow automation layer — the protocol, monitoring, and certification infrastructure that makes non-opioid recovery structured and sellable for surgery centers.
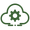
In January 2025, the FDA approved Journavx (suzetrigine), the first non-opioid analgesic in a new drug class in over 20 years. Developed by Vertex Pharmaceuticals, it selectively blocks NaV1.8 sodium channels in the peripheral nervous system — intercepting pain signals before they reach the brain without touching opioid receptors. No euphoria. No addiction pathway. No cognitive fog. In Phase 3 trials across roughly 2,200 patients, it delivered pain relief comparable to hydrocodone/acetaminophen with a milder side-effect profile (mainly nausea, constipation, headache, and dizziness) and zero evidence of dependence. The FDA gave it Breakthrough Therapy and Fast Track designations.
Then in early March 2026, the signal sharpened. Vertex released Phase 4 data from a multicenter study across aesthetic and reconstructive plastic surgeries showing 90.9% of patients remained opioid-free through the end of treatment (up to 14 days). Standard multimodal protocols typically keep fewer than 10% of patients opioid-free. That gap is massive, and it was generated in precisely the specialty where patients pay the most attention to their recovery experience.
Pharma will sell the molecule. Surgeons will adopt it. Patients will hear the promise. Very few people will build the system that makes the promise measurable, repeatable, and marketable.
The convergence
Three forces are hitting at the same time.
Regulatory push. The NOPAIN Act, enacted in 2023, requires CMS to provide separate Medicare reimbursement for qualifying non-opioid pain management products in hospital outpatient departments and ambulatory surgical centers. These temporary add-on payments run through December 31, 2027. The federal government is literally paying surgery centers extra to adopt non-opioid protocols — and that window is ticking.
Provider demand. Hospitals are already marketing opioid-free surgery as a patient-facing brand. UPMC Shadyside launched its Opioid-Free Surgical Pain Management Program in May 2024. Within six months, 109 patients enrolled, and 79 completed surgery without opioids. The program was patient-initiated — surgeons saw enough inbound demand that they built a formal pathway around it.
Commercial traction. Vertex reported over 550,000 Journavx prescriptions filled by year-end 2025, up from 20,000 in March. All three major PBMs now cover it. Over 170 million Americans have covered access. Vertex expects prescriptions to more than triple in 2026.
The molecule is moving. The policy is in place. Patient demand exists. What's missing is the operating system that makes non-opioid recovery structured and marketable for surgical centers.
What patients actually want
The sale isn't "non-opioid." That's a mechanism story. The consumer story is clear-headed recovery.
Patients want to wake up after surgery, go home, and heal without the haze, the nausea, or the quiet fear that their pain meds might become a problem. Suzetrigine acts peripherally, avoiding the CNS depression that causes classic opioid grogginess. The consumer promise — full mental clarity during recovery — actually holds up.
That distinction changes the business. A "non-opioid recovery concierge" is a compliance product. A "clear-headed recovery platform" is a premium care-experience product. The best surgery centers aren't selling procedures — they're selling outcomes and peace of mind.
Picture the patient's decision. You're choosing between two plastic surgery centers. One hands you a Vicodin script and says "call us if you have problems." The other enrolls you in a structured recovery program, monitors your pain daily, tracks your sleep and mobility, shows you your own recovery trajectory, and can demonstrate that 90%+ of their patients never needed opioids.
That second center closes the patient almost every time.
The business: Recovery OS
Recovery OS is a software-plus-protocol company for outpatient surgery centers — a micro SaaS for surgical recovery workflows. It helps ASCs, plastics groups, and procedure-focused clinics launch and run a branded opioid-sparing recovery pathway.
The product has four layers.

Unlock the Vault.
Join founders who spot opportunities ahead of the crowd. Actionable insights. Zero fluff.
“Intelligent, bold, minus the pretense.”
“Like discovering the cheat codes of the startup world.”
“SH is off-Broadway for founders — weird, sharp, and ahead of the curve.”